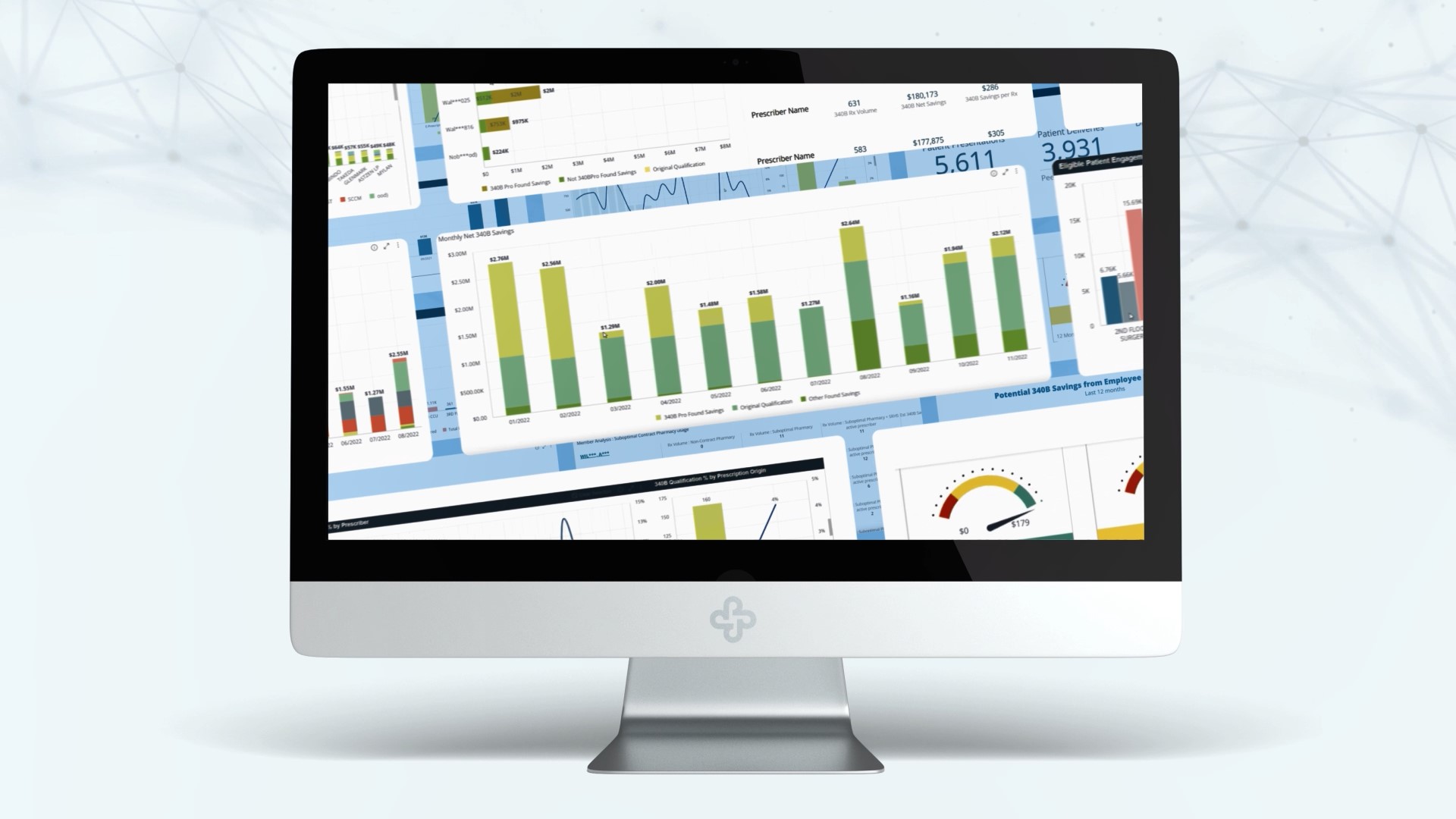
In the past week, no less than three articles have called attention to the restrictions drug manufacturers have imposed to evade their legal responsibility to honor 340B program prescription discounts — as well as the impact those restrictions have had on 340B-eligible nonprofit entities’ ability to serve their patients and communities. “Don’t believe drug companies: … Read More
340B ESP
Leveraging Partnerships With Drug Manufacturers To Overcome 340B Restrictions On Specialty Drugs
Since the advent of 340B ESP in 2020, drug manufacturers have imposed a raft of unlawful restrictions on 340B program savings. Nowhere have those restrictions been more punitive than with specialty pharmacy medications; drugs which typically represent the most expensive (and profitable) in manufacturers’ product lines. 340B restrictions from the drug manufacturers’ perspective That said, … Read More
The 2023 Guide To Implementing An Effective 340B Program
The 340B-program landscape has changed dramatically. For the 340B program’s first 28 years after its 1992 inception, the basic list of Best Practices for eligible entities (the 340B basics, if you will) remained largely unchanged — aside from the dramatically-increased importance of using specialized software. But since the June, 2020 introduction of 340B ESP — … Read More
Why 340B Hospitals Should Now Build Their Own Specialty Pharmacies
340B ESP and drug manufacturer restrictions are only part of the reason As we noted in a previous post, for 340B-eligible hospitals dealing with increasingly squeezed bottom lines, an in-house specialty pharmacy offers enormous savings and revenue potential. Some covered entities generate as much as 600% in specialty pharmacy revenue from 340B drugs as they … Read More
Manufacturers Accelerate Pace And Severity Of 340B Drug Pricing Restrictions
There’s an old saying that it’s better to be rich and guilty than poor and innocent. It’s a sad truth that accurately reflects the current state of 340B manufacturer restrictions, particularly in the wake of the January 30, 2023 decision by the U.S. Court of Appeals for the Third Circuit — which was largely in … Read More
Brick Walls: Dealing With 340B Manufacturer Restrictions.
A VytlOne Team Member Speaks Out. I joined VytlOne in early 2021, and I’ve been devoted to 340B program management full-time since early 2022. I’m no stranger to heavily regulated programs. Nearly my whole professional career has been in some aspect of public health — preclinical, clinical and now post-clinical. I’m currently fully devoted to … Read More
The Shame Of 340B ESP and Drug Manufacturer Restrictions
Making the rich richer at the expense of the poorest and most vulnerable. Why was the 340B program created? Nobody’s ever put it better than the Association of American Medical Colleges (AAMC): “Congress created the 340B Drug Pricing Program in 1992 to protect safety-net hospitals from escalating drug prices by allowing them to purchase outpatient … Read More
7 Steps For Overcoming 340B ESP & Other Pricing Restrictions
POST OUTLINE 1) Our process for recovering savings lost to 340B ESP Introduction / Overview Brief history of 340B-related manufacturer restrictions List of manufacturers currently imposing restrictions 2) How VytlOne overcomes 340B ESP restrictions on 340B pricing Establishing communications protocols Ensuring 340B ESP restores prices on schedule Recommendations for further reading Ensuring 340B ESP restores … Read More
How To Manage Your 340B Program Without Hiring More Staff
Two DOPs Deal With Operations, The 340B Program, And Drug Manufacturers’ Restrictions VytlOne recently organized an introductory Zoom meeting between the DOPs of two health systems we serve. Southwest Mississippi Regional Medical Center’s (SMRMC) Tiffany Poole earned her Doctorate of Pharmacy degree from the University of Mississippi in 2006. A VytlOne pharmacist since 2018, she … Read More
What Is Your ROI Potential With An Optimized 340B Program?
Introducing VytlOne’s 340B Program ROI Calculator The 340B program has often been described, and with good reason, as a black box. There are dozens of factors impacting an individual hospital’s relative success or failure in generating positive returns from a program. VytlOne has optimized dozens of 340B programs — generating over $500 million in savings … Read More